Timing often is happenstance, but with Earth Day arriving next week, word of high mercury levels in fish in national parks from Alaska to Colorado is particularly sobering. In some cases, the levels exceed limits OKed for human consumption.
One fish sampled for the study, taken from Yosemite National Park in California, had mercury levels of 950 parts per billion, "at which no human consumption is advised," said the report produced by the U.S. Geological Survey and the National Park Service.
All 21 parks included in the four-year survey, from Denali National Park and Preserve in Alaska to Rocky Mountain National Park in Colorado, had fish with varying levels of mercury. Concentrations ranged from 9.9 parts per billion to 1,109 parts per billion, with a mean of 77.8 parts per billion.
'Unfortunately, mercury and other forms of pollution don't recognize park boundaries. Even our nation's most pristine places like our national parks are vulnerable to toxic pollution," said Sarah Gaines Barmeyer, director of Conservation Programs for the National Parks Conservation Association in an email.
"When you visit national parks, you expect to see the best of what America has to offer, which should not include fish consumption advisories. This makes the USGS study particularly concerning. NPCA supports efforts to reduce mercury pollution from coal plants and other industries that contaminate park air and water.'
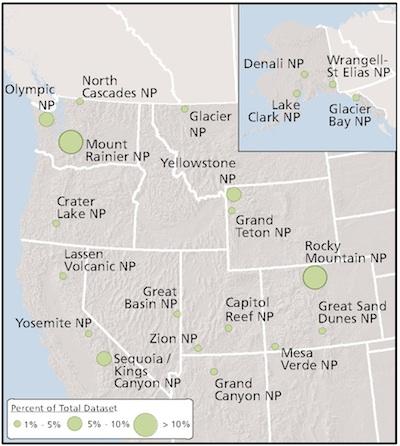
Spatial distribution of the 21 national parks sampled in this study. Size of circle represents percentage of total dataset.
While only 5 percent of the nearly 1,500 fish sampled had mercury concentrations exceeding a benchmark of 200 parts per billion that elicits toxic responses in fish, 35 percent of the samples had mercury levels above 90 parts per billion, high enough to impact some birds, and mercury levels in 68 percent of the fish "were above exposure levels recommended by the Great Lakes Advisory Group for unlimited consumption by humans," the study said.
"Zion, Capital Reef, Wrangell-St. Elias, and Lake Clark national parks all contained sites in which most fish exceeded benchmarks for the protection of human and wildlife health," the report's abstract noted. "This finding is particularly concerning in Zion and Capitol Reef national parks (in Utah) because the fish from these parks were speckled dace, a small, invertebrate-feeding species, yet their mercury concentrations were as high or higher than those in the largest, long-lived predatory species, such as lake trout."
Interestingly, just as the researchers found varying mercury levels from park to park, some fish samples taken within a single park had wide swings in the levels.
"The greatest range in site-specific geometric mean fish (total mercury) concentrations within parks occurred in Mount Rainier (in Washington state) and Rocky Mountain (in Colorado) national parks, where we measured approximately 11- and 8-fold differences, respectively, between the sites within each park with the lowest and highest (total mercury) concentrations," the researchers noted.
The sampling found that while Grand Teton (in Wyoming) and Great Basin (in Nevada) national parks "had the lowest proportion of fish exceeding the (Great Lakes Advisory Group) unlimited consumption threshold (18 percent), ... 100 percent of fish analyzed from Glacier National Park (in Montana) exceeded this threshold."
Overall, though, 14 of the 21 parks involved in the survey "did not have any fish in exceedance of the 300 parts per billion EPA fish tissue criterion," the report said. "Conversely, for those parks with fish that did exceed ecological and human health benchmarks, a substantial proportion of the fish had concentrations that were above those thresholds of concern."
Some fish in Wrangell-St. Elias National Park and Preserve and Lake Clark National Park and Preserve, both in Alaska, exceeded thresholds recommended by both the State of Alaska and the Environmental Protection Agency for unlimited human consumption by women and children, a Park Service release said.
State of Alaska human consumption thresholds start at 150 parts per billion. Lake trout, with average concentrations as high as 417 parts per billion, were found in one lake in Wrangell-St. Elias National Park and Preserve. The average concentration in lake trout in Lake Clark was 365 parts per billion. Mercury levels were low in Denali National Park and Preserve and Glacier Bay National Park and Preserve.

One of the brook trout sampled at Mount Rainier National Park./USGS, Collin-Eagles Smith.
Park Service officials in Alaska noted that "Lake trout are a relatively long-lived fish species at the top of the food web, and as such are more likely to accumulate mercury and other toxins over their lives than other species of fish. They are not consumed in great quantities by either sport or subsistence fishermen so the risk to people is likely low, but variable given location and frequency of fish consumption. The extent of the effects on fish-eating birds and mammals that depend on lake trout for survival is unknown."
A joint press release from the USGS and Park Service pointed out that the Park Service "is coordinating with state officials in the 10 study states regarding potential fish consumption advisories. State fish consumption guidelines consider both the risks associated with mercury exposure and the benefits of fish consumption, such as improved cardiac health from increased omega-3 fatty acid consumption or potential reduced intake of unhealthy fats due to food substitutions."
According to the paper, mercury is "among the most widespread" of environmental contaminants, coming from both natural sources (volcanic eruptions), and manmade sources (coal-fired power plants as well as historic and current mining activities). "more than 16 million lake acres and 1 million river miles are under fish consumption advisories due to mercury contamination in the United States, and 81 percent of all fish consumption advisories were issued because of mercury contamination," it notes.
The study, 'Mercury in fishes from 21 national parks in the Western United States ' Inter and intra-park variation in concentrations and ecological risk,' is available here. Within its pages you can find breakouts on the 21 individual parks.
Additional information on mercury pollution in national parks can be found here.


Comments
This is an interesting study with very interesting results. I wonder how in-park fish concentrations of mercury compare with concentrations in fish collected elsewhere? What about mercury concentrations in freshwater and salt water fish purchased from commercial markets? Also, I wonder why only mercury was sampled?
With the same effort invested to collect and sample fish for mercury, a much wider spectrum of toxic metals and radioactive substances could be analyzed, and in so doing, the national parks could function as ecological benchmarks for comparison to urban, suburban, and rural areas with higher potential for contamination of aquatic biota.
One way or another, this study demonstrates how difficult it is to protect natural ecosystems from external threats, especially those involving contamination of the atmosphere and subsequent deposition of toxic materials onto distant landscapes.
One reason Crater Lake has such low concentrations of mercury in its fish is because all water received by the lake is from direct precipitation and the lake's surface itself represents a rather small surface area for direct deposition of atmospheric contaminants.
=====
According to the paper, mercury is "among the most widespread" of environmental contaminants, coming from both natural sources (volcanic eruptions), and manmade sources (coal-fired power plants as well as historic and current mining activities). "more than 16 million lake acres and 1 million river miles are under fish consumption advisories due to mercury contamination in the United States, and 81 percent of all fish consumption advisories were issued because of mercury contamination," it notes.
=====
So obviously, we need to address containing volcano eruptions?
It would be interesting to know what percentage of mercury contamination in U. S. waters is believed to come from natural sources (such as volcanic eruptions) vs. human activities. I know concerns about mercury were a big issue several years ago following proposals to built several large, coal-fired power plants in Texas. As I recall, the coal-fired options were either greatly scaled back or dropped completely, in favor of natural gas.
An article on this topic from last fall in the Dallas Morning News says market forces (including cheaper natural gas) are causing a shift away from coal power, at least in Texas, and wind-generated power has become a larger player in Texas as well.
This article in Forbes last month says Texas set a new record for power from wind: On March 26, "close to 29% of the nearly 36,000 MW of electricity on the power grid at that moment" came from wind power.
No source of power is without some impacts on the environment, but at least wind power doesn't spread contaminants such a mercury, and as the Forbes article points out, alternatives to coal are becoming viable.
Looks like Rick is on to something.
http://www.sciencedaily.com/releases/2008/06/080629081932.htm
Seven tons from one vent of one volcano. US power plants generate in total about 53 tons.
Owen, i agree, EC the post was informative. Can you direct me to the site where the figure of 53 tons per year for the US from power plants was cited? Several years ago we did a toxic mineral study of 150 lakes in Yosemite National Park. We found significant levels of mercury in only one, Miller Lake on the PCT. In any case, it would be interesting to note world wide power plant mercury pollution if anyone has knowledge of that information.
rmackie
Here is one source for the 53 ton number.
http://www.epa.gov/mats/powerplants.html
Do a google search, there are plenty of sources for the world wide number. Overall it looks like anthropogenic sources are between 25 and 40% of all output with Asia being by far the largest contributor - 10x times the output of North America.
http://epa.gov/oia/toxics/mercury/mercury_context.html
Given how surprised about the magnitude of output of that single vent the scientist were in the prior article, my guess is that the above piece drastically underestimates the natural output.
That would suggest the source was local. If the source identified as was local thermal activity, should man try to reduce the leaching into the lake or let the fish die?
EC, thank you for the references. I am very uninformed on the issue, and certainly there is little that can be done about the natural process, volcanic eruptions, earthquakes, mega storms etc. But it does seem to me we can do much about the human activity contributing to the problem. It is not only the toxic metal issue, its the chemicals, pesticides, pollution and depletion of aquifers, surface water sources, air quality concerns including airborne contaminates, well the list is quite lengthly. Then there is the effort to protect species habitat and diversity. I think there are serious environmental issues, they should not be taken to lightly.